
- BAND GAP GROUND STATE VALENCE BAND CUT OFF WAVELENGTH SERIES
- BAND GAP GROUND STATE VALENCE BAND CUT OFF WAVELENGTH FREE
This can be true for the ground state and the excited state. In other words, n=0, v does not = 0 and r does not =0. Often, during electronic transitions, the initial state may have the electron in a level that is excited for both vibration and rotation. This is called fluorescence and can be detected in the spectrum as well.Įmbedded into the electronic states (n=1,2,3.) are vibrational levels (v=1,2,3.) and within these are rotational energy levels (j=1,2,3.). Once in the excited state, the electron has higher potential energy and will relax back to a lower state by emitting photon energy. If the wavelength of the incident beam has enough energy to promote an electron to a higher level, then we can detect this in the absorbance spectrum. 8.Often, during electronic spectroscopy, the electron is excited first from an initial low energy state to a higher state by absorbing photon energy from the spectrophotometer.Mismatch for Cells Connected in Parallel.
BAND GAP GROUND STATE VALENCE BAND CUT OFF WAVELENGTH SERIES
Solar Radiation Outside the Earth's Atmosphere.Although it is actually the liquid that moves, it is easier to describe the motion of the bubble going in the opposite direction. The concept of a moving "hole" is analogous to that of a bubble in a liquid. Thus, both the electron and hole can participate in conduction and are called "carriers". Consequently, the excitation of an electron into the conduction band results in not only an electron in the conduction band but also a hole in the valence band. The continual movement of the space for an electron, called a "hole", can be illustrated as the movement of a positively charged particle through the crystal structure. When this electron moves, it leaves behind another space. An electron from a neighboring atom can move into this empty space. The excitation of an electron to the conduction band leaves behind an empty space for an electron. However, the excitation of an electron to the conduction band will also allow an additional conduction process to take place.
BAND GAP GROUND STATE VALENCE BAND CUT OFF WAVELENGTH FREE
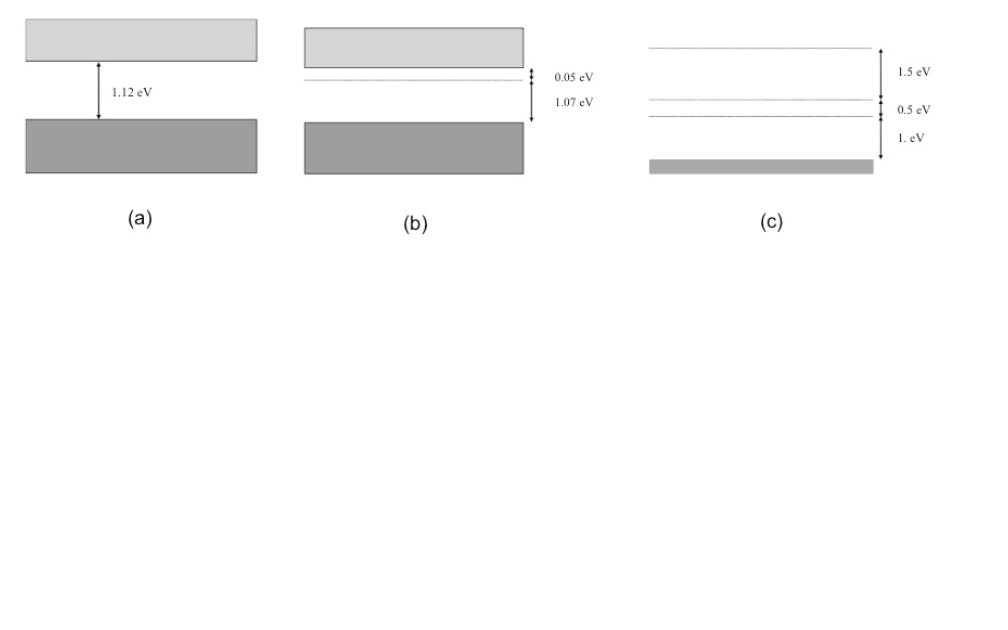
Once the electron becomes excited into the conduction band, it is free to move about the semiconductor and participate in conduction. Schematic of the energy bands for electrons in a solid. Therefore, the band gap is the minimum change in energy required to excite the electron so that it can participate in conduction. The band gap (E G) is the gap in energy between the bound state and the free state, between the valence band and conduction band. The lower energy level of a semiconductor is called the "valence band" (E V) and the energy level at which an electron can be considered free is called the "conduction band" (E C). The band structure of a semiconductor gives the energy of the electrons on the y-axis and is called a "band diagram". The band gap of a semiconductor is the minimum energy required to excite an electron that is stuck in its bound state into a free state where it can participate in conduction. This hole also participates in conduction. A hole is created where the electron was formerly bound.The band gap determines how much energy is needed from the sun for conduction, as well as how much energy is generated.When the band gap energy is met, the electron is excited into a free state, and can therefore participate in conduction.The band gap is the minimum amount of energy required for an electron to break free of its bound state.

